The UK Rare Diseases Framework, introduced in 2021, set out four pillars to improve the quality of care received by individuals with rare diseases:1
Over the past five years, England, Wales, Scotland and Northern Ireland have all published action plans aimed at turning these pillars into reality. In this final year under the existing framework, and as part of our Rare Diseases team’s activities for Rare Diseases Day, we reflected on the current UK rare disease landscape. We’ve identified policies and initiatives we feel have the greatest potential to improve equity in access to appropriate care for individuals with rare diseases. This is our summary of the hot topics to keep an eye on this year.
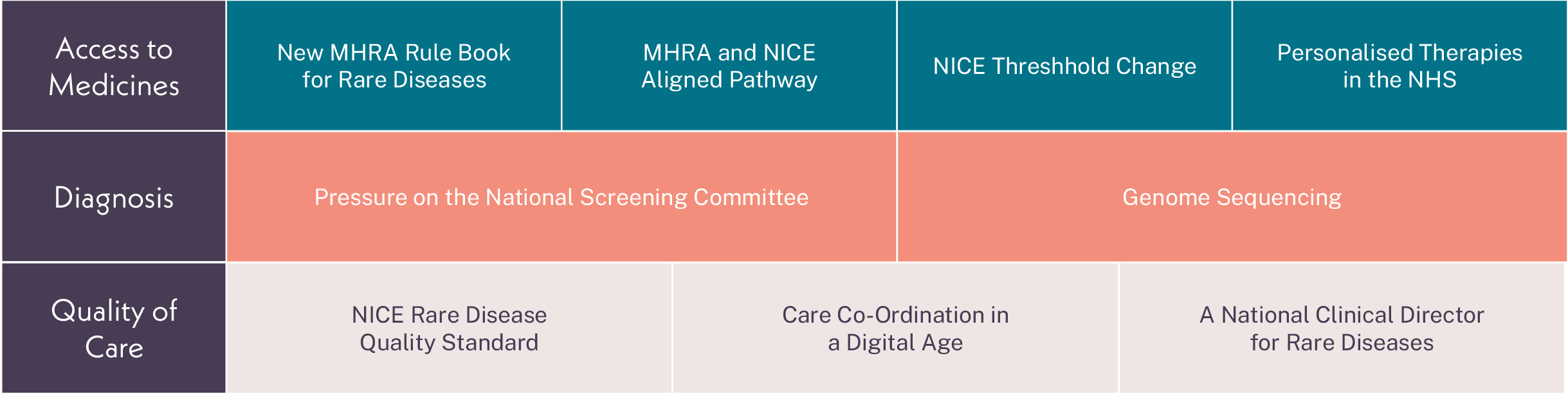
Abbreviations: MHRA: Medicines and Healthcare products Regulatory Agency (MHRA); NHS: national health service; NICE: National Institute for Health and Care Excellence.
Access to medicines is where some of the biggest movements are being seen. The Medicines and Healthcare products Regulatory Agency (MHRA) announced a planned overhaul of its approach to rare diseases, in a paper published last November.2 Their stated objective is to speed up the pathway for rare diseases from discovery to delivery. Current communications from the MHRA, such as their presentation at a recent UK parliamentary reception held by the UK BioIndustry Association (BIA), have been vague and non-committal. We are therefore keen to see the “new rulebook” that they will publish later this year. However, one bold idea floated has been a single approval for both clinical trial application and marketing authorisation.
When we discuss access to medicines, we are ultimately talking about ensuring that the right patients receive the right treatments as quickly as possible, and funding of medicines is a critical part of this. Whilst the MHRA is demonstrating that they are willing to be more flexible in their evidence requirements for rare diseases, we are not seeing a similar change in expectations from the National Institute for Health and Care Excellence (NICE), in the way they evaluate medicines for rare diseases. Will we therefore see an increasing divide in expectations from the MHRA and NICE?
On the 1st April 2026, the MHRA and NICE launched their new, optional pathway for parallel decision-making on licensing and value.3 The intention is to ensure that marketing authorisation and reimbursement decisions can be made simultaneously, creating faster access, by bringing NICE decision making earlier. The pathway will require very advanced preparations, with one requirement being registration of the medicine for UK national horizon scanning at least 3 years in advance of anticipated marketing authorisation. Manufacturers choosing this pathway may also need to submit to NICE in advance of MHRA, a notable change from typical sequencing, requiring companies to have a “NICE ready” evidence base earlier. This raises the question of how many companies will engage with this pathway and would be in a position to make their submission to NICE within the requisite timelines, considering the more extensive evidence requirements for NICE.
Additionally, whilst the NICE cost-effectiveness threshold change may generally improve the appeal of the UK market to manufacturers, it’s unlikely to have an impact on rates of rare disease therapies being reimbursed in the UK. Only a very small proportion of technologies routed to the Standard Technology Appraisal (STA) pathway would likely be re-evaluated as cost-effective under the new threshold.4 Reform of the NICE process was discussed at a RareRevolution webinar in November 2025, with stakeholders echoing that ‘tinkering around the edges’ of thresholds will not solve the problem of access for rare disease medicines. Instead, the BIA’s rare disease industry group (RDIG) are pushing for more radical reform such as an entirely separate appraisal route not based on cost-effectiveness.
It feels that the benefits of the MHRA changes will be mostly for individualised/personalised medicines, reflecting the recent milestones reached in this space. In February 2025, we saw the treatment of baby KJ with the first on-demand CRISPR gene therapy, demonstrating that this technology can be successfully used for ultra-rare, patient-specific, genetic causes of disease.5 In most cases, it is clinicians and academic groups driving personalised therapy development, rather than the pharmaceutical industry. At the International Society for Pharmacoeconomics and Outcomes Research (ISPOR) 2024 Europe conference, our US Head of Rare Diseases Kate Hanman chaired a panel entitled “Is It Time for More Non-Profits to Lead Reimbursement and Commercialisation of Gene Therapies to Promote Affordable and Sustainable Access?”. It certainly feels like we will continue to see this direction of travel in 2026.
Interestingly, an enquiry is now being launched into innovation in the NHS, with a focus on personalised medicine and artificial intelligence (AI) as examples of why the NHS adoption of innovations often fails.6 Whilst personalised medicines are exciting developments, many in the rare disease community have been struggling to access approved treatments, due to challenges with diagnosis, service delivery or reimbursement. Focusing more on these fundamental issues within the NHS and UK access pathways might be a better investment of resources to meet patient needs.
Developing innovative medicines is important, but early diagnosis is essential to ensure patients can benefit from these innovations, particularly when treatments need to be provided in the pre-symptomatic period. We’ve seen this exemplified in spinal muscular atrophy (SMA), a condition for which there are now many approved treatments, and where early diagnosis and treatment lead to better outcomes. The NHS newborn blood spot (NBS) screening does not currently include SMA, despite many other countries in Europe having been screening for SMA in newborns for many years and NHS Scotland rolling out SMA screening (in a research capacity) for every baby born from March 2026.7 With celebrity input into petitions, this will continue to be a hot topic, with increasing pressure being placed on decision-makers at the UK National Screening Committee (NSC).8 A recent update states that the NSC is “working with experts and partner organisations to look at how to make it easier to develop the evidence needed to make robust recommendations on the addition of more rare diseases to the NBS screening programme”.9
The NHS 10-Year Health Plan published last year had a strong focus on widening access to genomics and early screening.10 As part of the “sickness to prevention” pillar of the strategy, it cites that this would help to address the diagnostic odyssey for some rare conditions “reducing the time to diagnosis from 4 years to 3 months in some instances”. The Generation Study being run by Genomics England looks to be a key tool in realising this policy objective. This groundbreaking research study will look to sequence the genomes of 100,000 newborn babies, and in partnership with the NHS, will look to understand whether we can improve our ability to diagnose and treat genetic conditions through genome sequencing.11 As with everything in the NHS, successful implementation will be key – capacity is likely to be a key constraint for wider, routine testing. It is promising to hear that pilot centres for children and adults with undiagnosed rare conditions (‘syndrome without a name’ or SWAN) are being opened across the UK, to explore where genomics research could help these individuals and their families.12
The publication of NICE’s Rare Disease Quality Standard in February is another key milestone already reached in 2026, which Costello Medical’s Rare Diseases team were pleased to be able to comment on within the public consultation in January.13 This represents a step forward in terms of monitoring the quality of care provided to individuals with rare conditions, or those who are undiagnosed but are suspected to have a rare condition. It is a positive move in establishing consistent expectations across the UK healthcare system, establishing mechanisms for measuring quality of care, and setting expectations for commissioners and providers to aspire to. We were particularly heartened to see specific pillars around equitable access to recommended treatments (Quality Statement 7), requirements for a named HCP co-ordinating care (Quality Statement 5), and requirements for broader holistic care throughout the care pathway (Quality Statement 6). Together, the new quality standards promote continuity, accountability, and patient-centred care, key demands we consistently have heard from the rare disease community in projects we have participated in. The key now will be translating these quality standards into actions.
In line with this positive ambition for improved quality of care for individuals with rare diseases, in the run-up to Rare Diseases Day, a group of stakeholders (led by Biogen) initiated a call for a National Clinical Director for rare diseases.14 These are individuals with a vital role in driving improvements in care outcomes across the NHS. We have recently seen a National Specialty Lead for rare oncology be instated as part of the new Rare Cancers Act, and so it feels a logical next step to ensure similar leadership is in place for other rare conditions.15
More broadly, the new NHS 10-Year Health Plan set out a core objective to free up hospitals to be able to focus on “world-class specialist care”, which could indicate greater capacity for more complex rare conditions. The plan also set out an ambitious objective for 95% of people with complex health needs to have an agreed care plan in place by 2027. Routine care is frequently complex, multidisciplinary and can straddle primary, secondary, tertiary and national/supranational centres. Care plans may allow specialist HCPs to set the standard for care, for those HCPs with more limited experience of the condition (e.g. in primary or emergency care settings) to follow and may also be impactful for planning around key life transitions.
However, the NHS 10-Year Health Plan also placed significant emphasis on increasing the usage of AI and digital tools, such as the NHS app, electronic health records and virtual appointments. Will this drive towards digital lead to a less ‘individualised’ or ‘personalised’ approach, and impact the quality of care co-ordination? Or will increases in efficiency better allow NHS services to focus on the complex care co-ordination cases?
While digitisation and genomics hold promise, they risk ignoring fundamental needs such as addressing NHS capacity constraints. These areas require attention alongside more innovative shifts if equity in access to appropriate care for rare diseases is to improve meaningfully.
For the rare disease community, policy signals need to translate into tangible improvements. The UK Rare Diseases Framework formally ends in January 2027. Genetic Alliance will play a central role in supporting the development of a new policy framework for rare diseases, engaging with the community to shape this future policy. Key themes from their Rare Diseases Day report include a call for a UK-wide map of rare conditions (a comprehensive rare condition registry), closing the evidence gap through fair research investment and reform of evidence standards, system-wide accountability for rare conditions and embedding rare conditions into mainstream healthcare delivery.16 A multi-stakeholder summit is planned for Autumn 2026 to consolidate insights and shape next steps.
As Genetic Alliance and other stakeholders prepare a forward-looking framework, our Rare Diseases team will continue to monitor policy developments, engage with patients, industry and providers, and leverage these policies to drive practical change wherever we see opportunities.
References
If you would like any further information or advice on the themes presented above, please get in touch, or visit our Rare Diseases page to learn how our expertise can benefit you. Issy Newell (Head of Rare Diseases) and Faye Bolan (Senior Analyst), created this article on behalf of Costello Medical. The views/opinions expressed are their own and do not necessarily reflect those of Costello Medical’s clients or affiliated partners.
